6.2 Creation of the Spectra
As sample molecules exit the GC column and enter into the mass spectrometer, they encounter an energy source. For the purposes of this chapter, the source is an electron impact tungsten filament at 70 eV (Section 5.2.1.2a). Energy emitted from the source removes a single electron from a sample molecule. This is the most basic reaction and is illustrated with methanol below (Figure 6.1).
e- + CH3OH- ---> [CH3OH] ·+ + 2e-
After these products move through the mass spectrometer, the detector is only sensitive to the positively charged molecules and is not sensitive to any neutral or radical molecules. The detector transforms the number of molecules into an electrical signal, and a computer or integrator translates the individual signal peaks into a bar graph. The abundance of each cation is plotted as a function of a molecule’s mass divided by its charge (m/z) on a bar graph (Figure 6.1). Since almost all of the fragments detected by the GC-MS have only a single positive charge, m/z is also a measurement of the molecules’ mass.
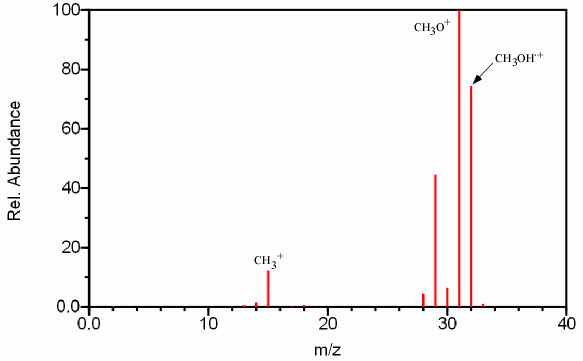
Figure 6.1 Mass Spectrum of Methanol. Spectra from the NIST/EPA/NIH Mass Spectral Library. Reprinted with permission from NIST.
Notice that there are more bars on the graph than just the total molecular mass of the sample molecule. These other peaks are the result of the cleavage of various bonds in the original sample molecule. The resulting fragments provide information about the original structure of the sample molecule like pieces of a puzzle (Section 6.8). Since the energy of the source exceeds the ionization energy of the sample molecule, the excess energy that is not utilized in the removal of a single electron is distributed over various electronic, vibrational, and rotational degrees of freedom (Section 6.6). Fragmentation occurs when the vibrational energy exceeds the activation energy of the cleavage of that particular bond (Section 6.6). This feature allows the instrument to distinguish between compounds with the same molecular mass. The major fragments for methanol (Figure 6.1) can be attributed to the following reactions.
[CH3OH-] ·+ --->CH3O+ + H ·
[CH3OH-] ·+ ---> CH2O+ + H2
[CH3OH-] ·+ ---> CH3+ + · OH
The two most important peaks in any mass spectrum are the base peak and the molecular ion peak. The base beak is the largest peak in the spectrum. In the case of methanol, the base peak is the peak at m/z 31 corresponding to the CH3O+ fragment. Since the absolute height of any peak is dependent on the concentration of the sample, the other peaks in the spectrum are referenced as a percentage of the base peak and referred to as relative abundance. This normalization of peak heights greatly aids in identification of fragmentation pattern and thus analyte identification.
The molecular ion peak (also referred to as the parent peak) corresponds to an analyte molecule that has not undergone fragmentation. In Figure 6.1, the molecular ion peak is caused by the [CH3OH-]·+ ion and corresponds to m/z 32. The molecular ion peak is often referred to as the [M+] ion. The molecular ion is used as a reference point in identifying other fragments. For example, the peak corresponding to m/z 15 is referred to as both [M – OH] and/or [M – 17].
| Frank's Homepage |
©Dunnivant & Ginsbach, 2008